- All Drug Categories
- Home
- Anti-cancer Drugs
- Healthcare Products
- Medical Information
- Statement
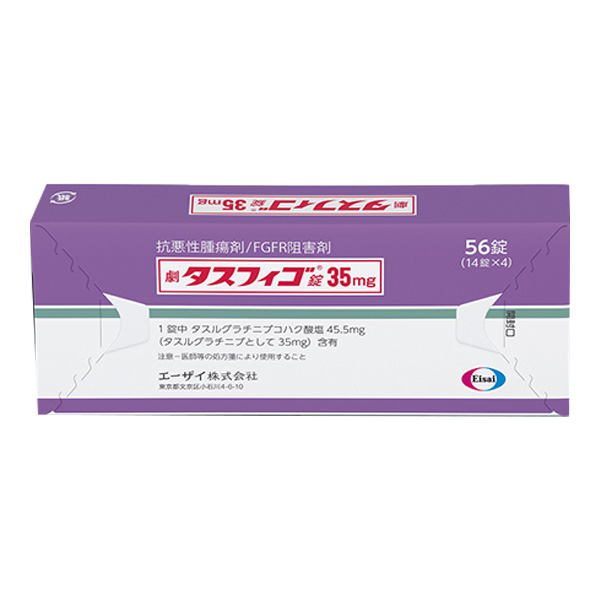
All Names: Tasfigo、tasurgratinib succinate、 琥珀酸他舒格替尼、タスフィゴ
Indications:Adult patients with unresectable biliary tract cancer with FGFR2 fusion gene positive progression after chemotherapy.
Manufacturer:Eisai, Japan
Customs Clearance Procedure:If the customs requires the package for customs clearance, please pay the customs clearance fee according to the content of EMS SMS and customs regulations.
Succinate sulfatinib is a novel oral tyrosine kinase inhibitor with selective inhibitory activity against FGFR1, FGFR2, and FGFR3, inhibiting tumor growth by suppressing and blocking these signals.
1、 Drug name
1. Common name: Tasurgratinib Succinate
2. Product Name: Tasfigo Tablets
3. English name: TasurgratinibSuccinate
2、 Indications
FGFR2 fusion gene positive, incurable biliary tract cancer used for the treatment of disease progression after cancer chemotherapy.
3、 Specifications and characteristics
1. Specification: 35mg per tablet.
2. Appearance: Yellow film coated slices.
3. Packaging specifications: 56 pieces/box (PTP packaging).
4、 Main components
Active ingredients: Each tablet contains 45.5mg of tasugtinib succinate (equivalent to 35mg of tasugtinib)
5、 Usage and dosage
1. Conventional dose: 140mg (4 tablets) once daily for adults, taken orally on an empty stomach.
2. Step by step reduction: When adverse reactions occur and require reduction, adjust in the following order: 140mg → 105mg → 70mg → 35mg → discontinuation.
6、 Dose adjustment and adverse reaction management
1. Hyperphosphatemia:
Blood phosphorus levels of 5.5-7.0mg/dL: Administer dietary therapy or phosphorus lowering medication.
Blood phosphorus levels ranging from 7.1 to 9.0 mg/dL: Administer phosphate lowering therapy, and re administer the medication after recovery to ≤ 7.0 mg/dL, with a reduction of one step.
Blood phosphorus ≥ 9.1mg/dL: Administer phosphate lowering therapy, suspend medication, and reduce dosage by one step after recovery to ≤ 7.0mg/dL before administering again.
2. Corneal or retinal damage (grade 2 or 3): Suspend medication and reduce dosage by one step after recovery to ≤ grade 1 before administering again; Level 4 patients will permanently discontinue medication.
3. Hematological toxicity (Grade 3): Suspend medication until it returns to ≤ Grade 2 before administering again; Suspend medication for level 4 patients until it returns to ≤ level 2, and then re administer medication.
4. Other adverse reactions (intolerable grade 2 or 3): reduce by one step; For level 4 patients, reduce the dosage by one step and then re administer the medication.
7、 Medication precautions
1. Medication time
Fasting administration: To avoid food affecting the efficacy of the medication, it is advisable to avoid taking it within 1 hour before meals to 2 hours after meals.
A high-fat diet can reduce peak drug concentrations and total exposure.
2. Handling of missed services
If missed, it should be taken as soon as possible when remembered.
If it is close to the next medication time, skip the missed dose and take the next medication according to the original plan.
Do not double the dosage to make up for missed doses.
3. Post vomiting treatment
If vomiting occurs after taking medication, there is no need to take it again.
Just wait for the next medication to be taken at the original dosage.
4. PTP packaging precautions
When taking medication, the tablets should be removed from the PTP board and taken.
If the PTP plate is accidentally swallowed, the sharp edge may puncture the esophageal mucosa, causing serious complications such as perforation and mediastinitis.
8、 Medication for special populations
1. Patients with liver dysfunction: This product is mainly metabolized in the liver, and the blood drug concentration may increase in patients with liver dysfunction. Clinical trials for patients with liver dysfunction have not yet been conducted.
2. Women with fertility: Effective contraceptive measures should be taken during medication and within 6 days after discontinuation.
3. Male patients should use barrier contraception methods such as condoms during medication and within 6 days after discontinuation.
4. Pregnant women: Use only when the benefits of treatment outweigh the risks. Animal experiments have shown that teratogenicity can occur at doses below clinical exposure.
5. Breastfeeding women: It is recommended to avoid breastfeeding as medication may enter breast milk and the baby may experience serious adverse reactions.
6. Children: Clinical trials for children have not yet been conducted, and safety and efficacy have not been established.
9、 Adverse reactions
1. Common adverse reactions (incidence rate ≥ 20%)
Hyperphosphatemia - the most common serious adverse reaction.
Nail damage, palm sole redness, swelling, and sensory impairment syndrome, diarrhea, paw inflammation, intraoral inflammation, taste disorders, fatigue, thrombocytopenia, leukopenia, and neutropenia.
2. Serious adverse reactions
Hyperphosphatemia: Regular monitoring of blood phosphorus and corresponding treatment are required.
Retinal detachment (8.7%): including serous retinal detachment, immediate ophthalmic treatment is required when there is blurred vision, floaters, visual field defects, flashes, or decreased vision.
3. Other common adverse reactions
Eyes: dry eye syndrome, keratitis, corneal epithelial defects, macular edema.
Gastrointestinal tract: dry mouth, constipation, nausea, vomiting, abdominal pain.
Skin: hair loss, dry skin, rash, itching.
Laboratory tests: Elevated AST, ALT, ALP, bilirubin, and creatinine levels.
10、 Contraindications
Patients with a history of allergies to any ingredients of this product are prohibited from using it.
11、 Drug interactions
1. CYP3A inducers (such as rifampicin): can reduce the blood concentration of tacrolizumab, and caution should be exercised when used in combination.
2. Proton pump inhibitors (such as rabeprazole): No significant effects were found and can be used in combination.
3. In vitro studies suggest:
Hesugtinib and its active metabolite M2 are substrates of P-gp.
Tachibatinib inhibits CYP3A, CYP4F12, and MATE1, while inducing CYP1A2, CYP2B6, and CYP3A.
He/she may inhibit P-gp and BCRP.
12、 Storage method
1. Store at room temperature and avoid moisture.
2. Store out of reach of children.
3. Store in PTP packaging and remove from the plate when taking.
13、 Manufacturer
Manufacturer: Eisai Corporation (Eisai Co., Ltd.).
14、 Other important information
1. Risk management: It is necessary to develop and strictly implement a drug risk management plan.
2. Expiration date: Please refer to the expiration date indicated on the packaging box. Expired drugs cannot be used.
Tasfigoinformation