- All Drug Categories
- Home
- Anti-cancer Drugs
- Healthcare Products
- Medical Information
- Statement
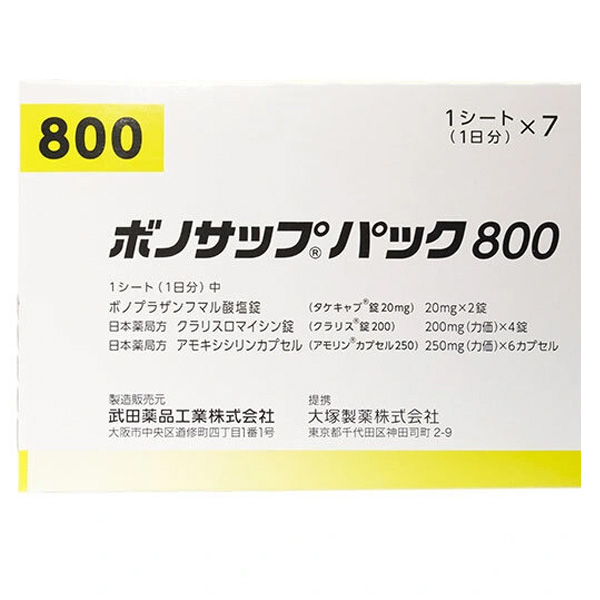
All Names: Vonosap Pack、Vonoprazan Fumarate, Amoxicillin Hydrate and Clarithromycin Combination Package、沃诺拉赞/阿莫西林/克拉霉素制剂组合包装、幽门螺旋杆菌根除治疗药
Indications:Suitable for treating infections caused by Helicobacter pylori that are sensitive to amoxicillin and clarithromycin. Specific indications include: gastric ulcer, duodenal ulcer, gastric MALT lymphoma (mucosa associated lymphoid tissue lymphoma), immune thrombocytopenia, Helicobacter pylori infection in the stomach after endoscopic treatment of early gastric cancer, and Helicobacter pylori infected gastritis.
Manufacturer:Takeda,Japan
Customs Clearance Procedure:If the customs requires the package for customs clearance, please pay the customs clearance fee according to the content of EMS SMS and customs regulations.
The VonosapPack, developed by Takeda Pharmaceutical Co., Ltd. in Japan, is a compound combination formulation specifically designed for the eradication of Helicobacter pylori.
1、 Drug name
1. Product Name: VONOSAP 800 ® Pack400&800)
2. Domestic reference translation: Wonosapubao 800
2、 Indications
1. Used to treat Helicobacter pylori infection that is sensitive to amoxicillin and clarithromycin.
2. Specifically applicable to Helicobacter pylori infection accompanying the following diseases:
Gastric ulcer, duodenal ulcer, gastric MALT lymphoma, immune thrombocytopenia, gastric cancer after endoscopic treatment, Helicobacter pylori infection gastritis.
3、 Specifications and characteristics
This product is packaged in combination, and the daily dose (1 tablet/pack) includes the following three formulations:
Poop ノサイプパイク 800 (Wonosap Pack 800):
1. Vorosuvate 20mg x 2 tablets.
2. Amoxicillin capsules 250mg x 6 capsules.
3. Clarithromycin tablets 200mg x 4 tablets.
4、 Main components
1. Vonohrase tablets: The active ingredient is Vonohrase fumarate salt.
2. Amoxicillin capsules: The active ingredient is amoxicillin hydrate.
3. Clarithromycin tablets: The active ingredient is clarithromycin.
5、 Usage and dosage
Please follow the doctor's advice for detailed usage and dosage.
6、 Dose adjustment
1. Patients with renal dysfunction:
The blood drug concentrations of Vorosuvastatin and clarithromycin may increase.
Patients with high renal dysfunction are prohibited from using this combination packaging as it is not possible to adjust the dosage of each preparation.
2. Patients with liver dysfunction:
The blood drug concentration of Vorosuvate may increase.
Clarithromycin may exacerbate liver dysfunction.
7、 Medication precautions
1. Usage: The three medications must be taken simultaneously according to their intended dosage.
2. Storage method: Store at room temperature. After opening, attention should be paid to moisture-proof storage (this product uses a high moisture-proof inner bag to ensure quality).
3. Regarding cases of missed doses, vomiting, etc.: It is necessary to strictly follow the doctor's instructions to take the medication in a timely and quantitative manner, and complete the entire course of treatment. If there is a missed dose or vomiting, please consult a doctor or pharmacist. Do not supplement or double the dose without authorization.
8、 Medication for special populations
1. Pregnant women: Use only when the benefits of treatment outweigh the risks. The relevant components have been observed to have fetal toxicity in animal experiments.
2. Breastfeeding women should consider both the benefits of treatment and breastfeeding before deciding whether to continue or stop breastfeeding. All ingredients can enter breast milk.
3. Children: Clinical trials targeting children have not yet been conducted, and safety is unknown.
4. Elderly people: Physiological functions (liver and kidney function) usually decline, and side effects (especially caused by amoxicillin) are more likely to occur. Blood concentrations of clarithromycin may remain high.
5. Comorbidity patients:
Individuals with a history of allergy to penicillin or cephalosporin antibiotics should use with caution (contraindicated for those allergic to amoxicillin).
Individuals or relatives with allergies such as bronchial asthma, rash, urticaria, etc. should use with caution.
Use with caution in patients with heart disease or hypokalemia (which may cause QT prolongation, ventricular tachycardia, etc.).
9、 Adverse reactions
1. Major adverse reactions:
Related to Vorosuvate: shock, allergic reactions, pancytopenia, granulocytopenia, leukopenia, thrombocytopenia, liver dysfunction, toxic epidermal necrolysis, mucocutaneous ocular syndrome, and erythema multiforme.
Amoxicillin related: shock, allergic reactions, acute coronary syndrome associated with allergic reactions, drug-induced gastroenteritis syndrome, toxic epidermal necrolysis, mucocutaneous ocular syndrome, severe kidney damage such as acute kidney injury, neutropenia, thrombocytopenia, pseudomembranous colitis, liver dysfunction, jaundice, interstitial pneumonia, aseptic meningitis.
Clarithromycin related: shock, allergic reactions, QT prolongation, ventricular tachycardia, ventricular fibrillation, fulminant hepatitis, liver dysfunction, jaundice, liver failure, thrombocytopenia, pancytopenia, hemolytic anemia, leukopenia, granulocyte deficiency, toxic epidermal necrolysis, mucocutaneous ocular syndrome, PIE syndrome, interstitial pneumonia, pseudomembranous colitis, hemorrhagic colitis, rhabdomyolysis, convulsions, acute kidney injury, tubulointerstitial nephritis, IgG vasculitis, drug-induced hypersensitivity syndrome.
2. Other side effects: Common ones include diarrhea, abnormal taste, etc.
10、 Contraindications
The following patients are prohibited:
1. Individuals with a history of allergies to any ingredients in this packaging.
2. Patients currently taking the following medications: Azanavir sulfate, Lipivirine hydrochloride, Pyrmozide, Ergotamine tartrate/anhydrous caffeine/isopropylantipyrine, Dihydroergotamine methanesulfonate, Suvorosen, Daliresen hydrochloride, Warnoxacin hydrate, Lometapine methanesulfonate, Tadalafil (for pulmonary arterial hypertension), Ticagrelor, Ibrutinib, Ivaprepitine hydrochloride, Vinaclat (for specific leukemia/lymphoma dose escalation periods), Rulaxone hydrochloride, Anamorilin hydrochloride, Finalidone, Esaconazole sulfate, Vorosporine, Marvacatine.
3. Patients with liver or kidney disease who are currently taking colchicine.
4. Patients with infectious mononucleosis have a higher risk of developing erythematous rash when using amoxicillin.
5. Patients with severe renal dysfunction.
11、 Drug interactions
1. Von Nora Sheng:
Taboo combination therapy: It is contraindicated with multiple drugs, mainly by affecting the pH value in the stomach and reducing the absorption of certain drugs (such as atazanavir and rivavirin).
Attention should be paid to combination therapy: Combination therapy with CYP3A4 inhibitors (such as clarithromycin) can increase the concentration of voxetine in the blood; Combined use with CYP3A4 inducers (such as rifampicin) can reduce the blood drug concentration of vonola; Combined use with digoxin may enhance its effects; Combined use with itraconazole, gefitinib, etc. may weaken their effects.
2. Amoxicillin:
May enhance the effect of warfarin potassium (inhibit intestinal vitamin K production).
May weaken the effectiveness of oral contraceptives (inhibiting enterohepatic circulation).
Combined use with probenecid can increase the blood concentration of amoxicillin.
May enhance the side effects of methotrexate.
3. Clarithromycin:
Taboo combination: Combining with many drugs that are mainly metabolized by CYP3A (such as pimozide, ergotamine, statins, colchicine, etc.) can significantly increase the blood drug concentration of these drugs, leading to serious adverse reactions.
Attention should be paid to combination therapy: When used in combination with digoxin, carbamazepine, theophylline, cyclosporine, etc., it can increase the blood concentration of these drugs; Combined use with rifampicin, efavirenz, etc. may reduce the self blood concentration of clarithromycin.
12、 Storage method
1. Storage conditions: Store at room temperature.
2. Validity period: 3 years.
3. Attention: Please pay attention to moisture prevention after opening, as the inner bag has high moisture resistance to maintain quality.
13、 Manufacturer
Takeda Pharmaceutical Industry Co., Ltd.
Vonosap Packinformation